When Brain–Body Networks Stop Adapting
Stress, pain, poor sleep, and mental fatigue are not just surface symptoms, they are signs that the autonomic nervous system (ANS) has lost its flexibility.
Normally, the ANS switches effortlessly between “fight-or-flight” and “rest-and-repair.” But chronic stress can lock the system in high alert, causing:
- Persistent sympathetic overdrive
- Elevated cortisol and inflammation
- Disrupted sleep architecture
- Reduced cognitive performance and slower recovery
The real problem isn’t just stress itself, it’s maladaptive brain–body signalling that stops the system from resetting.
The Vagus Nerve: The Key Pathway Back to Balance
The vagus nerve is the primary communication route between the brainstem and vital organs. It regulates heart rate, breathing rhythm, digestion, immune response, and emotional state.
What makes it uniquely accessible is that one branch of the vagus nerve, the auricular branch (ABVN), travels to the ear, specifically the cymba conchae and tragus regions. This is the only place on the surface of the body where vagal afferent fibres can be stimulated non-invasively.
Research shows that low vagal tone is linked to:
- Poor stress resilience
- Increased cardiovascular risk
- Higher rates of anxiety, depression, and chronic disease
Strengthening vagal signalling has become one of the most promising ways to restore autonomic balance, lower physiological stress load and support long-term health.
SECTION 3
How We Target and Retrain These Networks
Stimulating the vagus nerve is not new but doing it with precision is what drives real change.
SONA focuses on neuroplasticity, the brain’s ability to rewire itself. We deliver stimulation designed to do more than activate vagal fibres; we use it to retrain brain networks over time.
This is structured neural training, not just passive stimulation, gradually restoring healthy network dynamics session by sesson.
Time-locked delivery
Stimulation is synchronised with heart rhythm and breathing cycles, creating an optimal “learning window” for the nervous system.
Sensory pairing
Stimulation is combined with sound and breath guidance, strengthening synaptic plasticity through Hebbian learning (“what fires together, wires together”).
Optimised parameters
We use waveforms supported by peer-reviewed evidence to maximise safety and efficacy.
SECTION 5
What the Evidence Shows
Our own published research and other peer-reviewed studies support vagus nerve stimulation as a therapeutic pathway for:
These findings show that targeted vagal engagement is a multi-system intervention, improving both mental and physical health outcomes.
The Next Frontier: Adaptive, Closed-Loop Recovery
The field of neuromodulation is rapidly moving from open-loop stimulation (same dose for everyone) to personalised, biomarker-driven interventions.
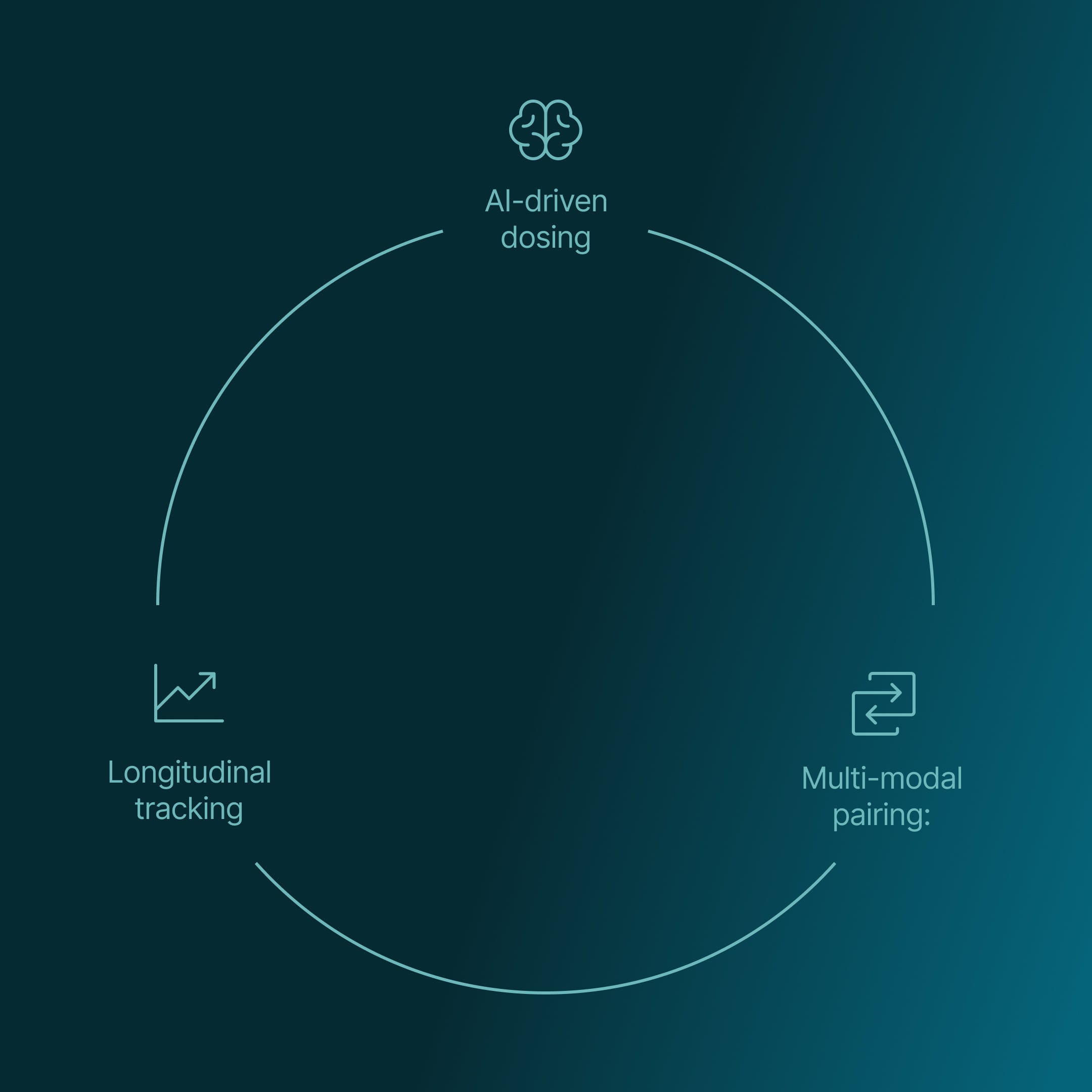
SONA is at the forefront of this transition, developing:
- AI-driven dosing: Adjusting stimulation in real time based on individual physiology
- Multi-modal pairing: Integrating sound, breath, and behavioural inputs for faster network reorganisation
- Longitudinal tracking: Building a data-driven picture of each user’s nervous system over time
This is what we call precision recovery, a tailored approach that trains the nervous system back to health.
THE RESULTS
What you can expect in Month 1
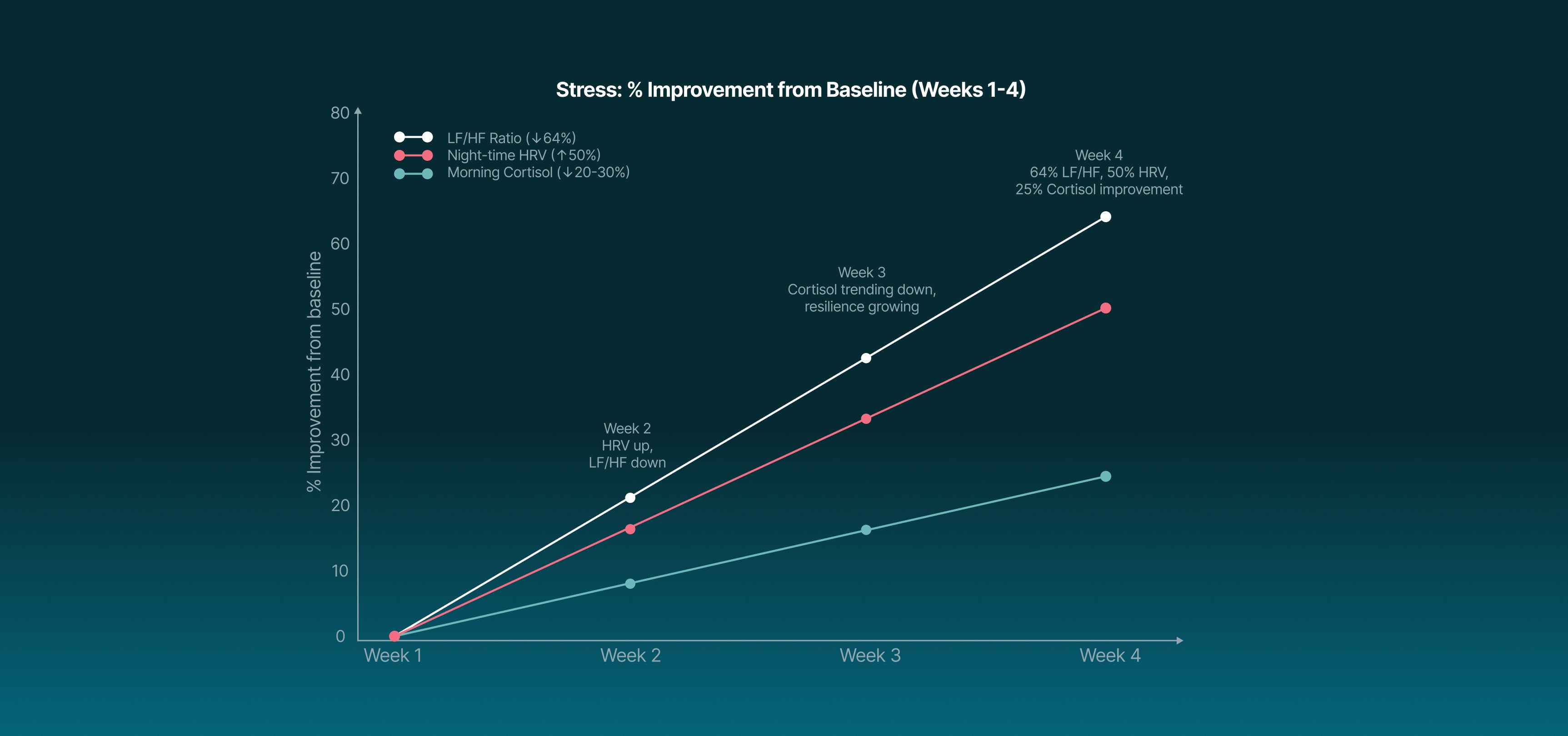
taVNS rebalances the autonomic nervous system, reducing sympathetic overdrive and increasing parasympathetic activity.
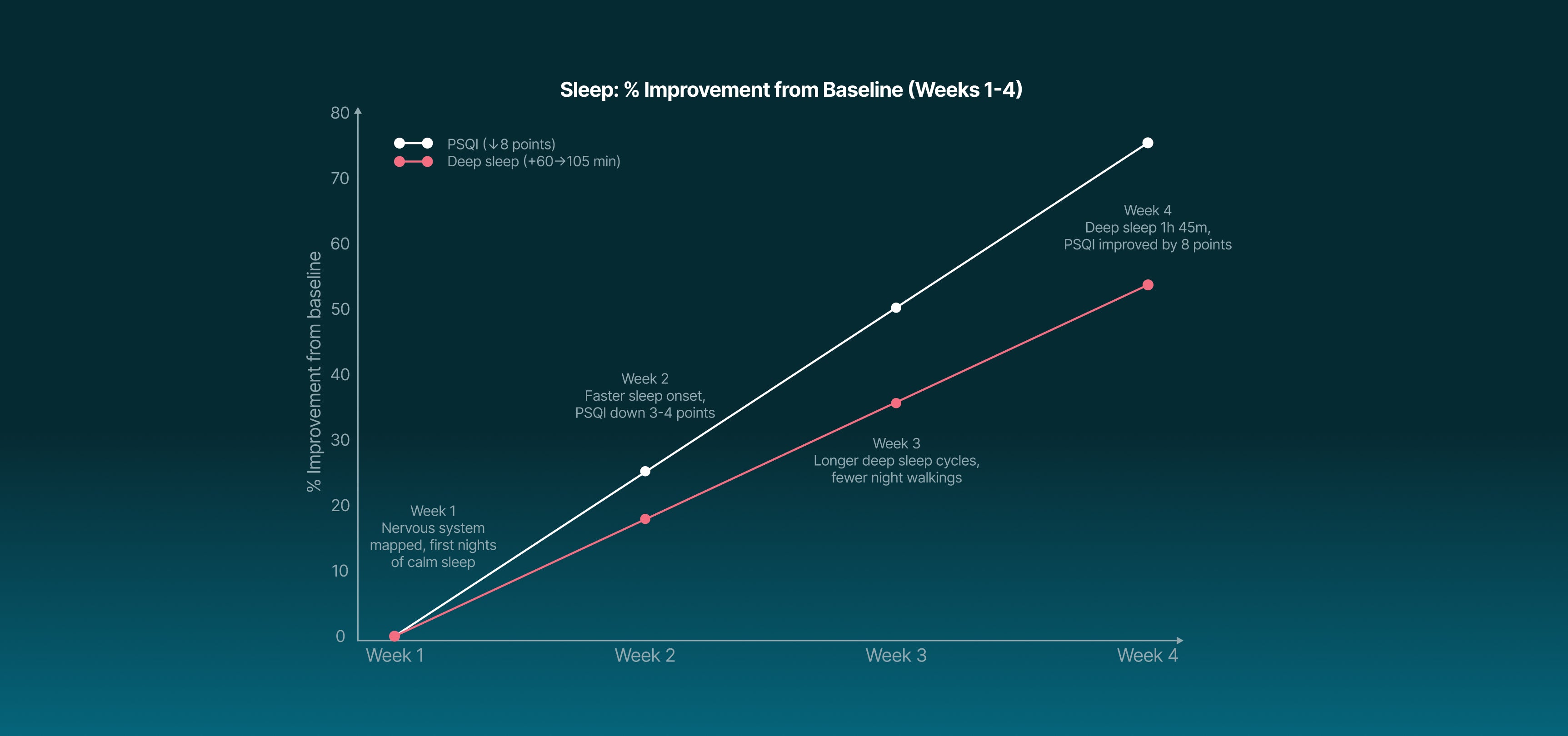
taVNS reduces overactive connections between the basal forebrain and visual, sensorimotor & medial PFC regions, helping the brain power down when you want to sleep.
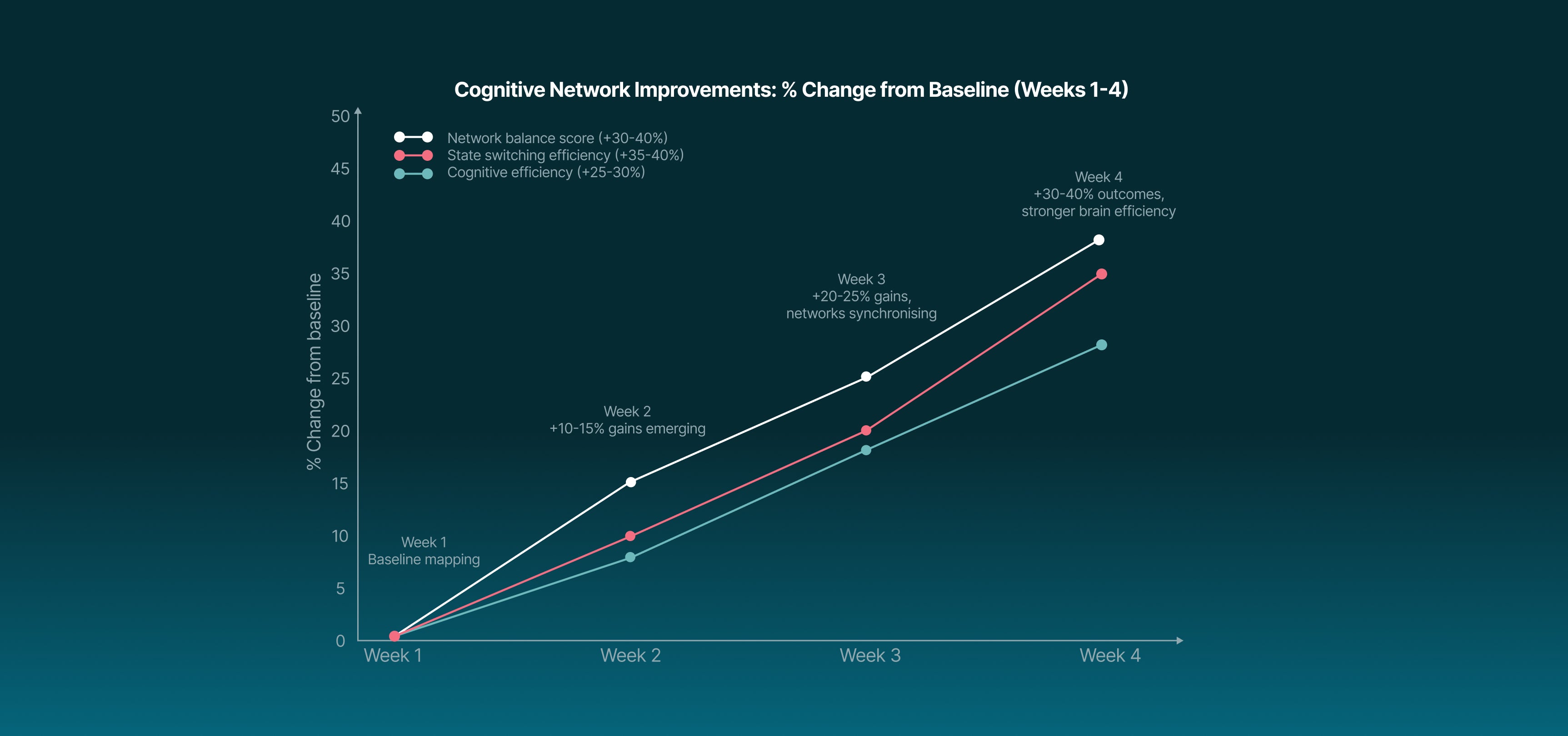
taVNS strengthens prefrontal and attentional network connectivity while dampening hyperactive default network mode activity, improving task engagement, network switching, and sustained attention.
End of month 1 snapshot:
HRV up to 2× baseline.
Deep sleep +30–40 min per night.
Sharper focus & faster reaction times.
Outcome: You’ve built a calmer, more resilient nervous system and you’re unstoppable.
Key Studies
Stress & Autonomic Regulation: Cuberos‑Paredes 2025
Design: 12‑subject single‑blind taVNS vs sham during Mental Arithmetic Stress Test
Key Findings: Cortisol rise during stress reduced by >50 %; HRV LF/HF suppressed; psoriasis case showed 40 % daytime cortisol reduction after 3‑month home use
Stress & Autonomic Regulation: Sanchez‑Perez 2023
Design: 19 healthy adults; four‑way crossover (taVNS, tcVNS, tMNS, sham)
Key Findings: taVNS shortened LVETI (↑ vagal tone) and raised PEP/LVET, indicating sympathetic withdrawal under cognitive & cold‑pressor stressors
Stress & Autonomic Regulation: Yoshida 2025
Design: 15 volunteers; randomised exercise stress test ± taVNS
Key Findings: Peak HR fell ~4 bpm at VO₂‑max; faster vagal re‑entry in recovery, negligible BP change, ideal for athletic use
Stress & Autonomic Regulation: Le Roy 2023
Design: 44‑person single vs triple‑session design
Key Findings: Three sessions/week increased HRV, cognitive throughput, and sleep efficiency; magnitude gated by baseline parasympathetic reserve
Sleep: Wu, Y. 2022
Design: Randomised, double-blind study with 30 patients diagnosed with primary insomnia
Key Findings: After one month, the active group saw greater improvement in sleep quality (73% vs. 27%) with no side effects. The results suggest taVNS is a safe and promising insomnia treatment
Sleep: Zhang, S. 2024
Design: Randomised trial - 68 patients with chronic insomnia
Key Findings: Found that taVNS significantly improved sleep in insomnia patients, reducing symptoms nearly twice as much as placebo (-8.2 vs -3.9 points). Benefits lasted 5 months and also helped mental health and fatigue, with no side effects.
Sleep: Yeom, J. 2025
Design: Clinical trial tested a non-invasive ear stimulation therapy (taVNS) on 40 patients with chronic insomnia
Key Findings: taVNS significantly improved sleep quality, insomnia severity, and overall quality of life compared to sham. These findings establish taVNS as a safe and effective non-invasivetherapy for managing insomnia, with potential for broader clinical applications.
Sleep: Jackowska, M. 2022
Design: Randomised, single-blind trial with 68 community-dwelling adults
Key Findings: While tVNS improved sleep quality in active groups, its efficacy over sham remains inconclusive. Larger, longer-term studies with objective measures are needed to clarify its therapeutic potential for non-clinical sleep complaints.
Cognitive Performance: Borges, U. 2020
Design: Vagally mediated heart rate variability parameters were measured in 32 participants
Key Findings: The results indicate for the first time (a) that tVNS can increase cognitive flexibility in a set-shifting paradigm, and (b) that tVNS may exert a stronger effect on cognitive flexibility than inhibition.
Cognitive Performance: Colzato LS, Ritter SM, Steenbergen L. 2018
Design: Tested whether gentle ear stimulation (taVNS) could boost creativity in 80 healthy adults
Key Findings: Results showed that active taVNS improved divergent thinking (generating many unique ideas, like brainstorming uses for a brick) compared to sham stimulation.
Cognitive Performance: Llanos, F. 2020
Design: 3 groups of 12 people
Key Findings: The researchers found that pairing subtle, imperceptible electrical pulses with easier-to-learn tones significantly improved learning speed and accuracy compared to no stimulation or stimulation paired with harder-to-learn tones. This non-invasive method could offer a promising way to enhance language learning in adults by targeting the right sounds at the right time.
Cognitive Performance: Ferstl M, Teckentrup V, Lin WM, et al. 2022
Design: 32 adults completed cognitive tasks while measuring their heart rate variability
Key Findings: The findings suggest taVNS may selectively enhance certain aspects of flexible thinking, but not broader mental control. This challenges theories about how vagus nerve stimulation affects cognition. Future research should explore why taVNS helps some mental processes but not others.
Cognitive Performance: Fischer, R. 2018
Design: How taVNS affects the brain's response to conflict called cognitive control
Key Findings: Found that taVNS improves conflict adaptation, boosting performance and altering N2 activity, likely due to the locus coeruleus-noradrenaline (LC-NE) system's involvement.
MindSpire & Partnered Research Pipeline
Our in‑house programme (MindSpire Labs) and academic collaborations extend taVNS into translational domains:
Tragus‑based stress reduction pilot
25 Hz, 200 µs: LF/HF down 64 %; α‑frontal EEG asymmetry shift toward relaxation in 5/5 subjects
30‑30 HRV biomarker study
Feasibility of HRV decline as pre‑symptomatic COVID signal; validated by subsequent large datasets
ReSONAnce Breathing Assessment (n = 51)
Identified 5 bpm as population‑mode reSONAnce, now embedded in SONA’s breath‑sync protocol .
TinnSpire (taVNS + sound) & Parkinson’s dual‑mode (taVNS+GVS)
Multi‑session feasibility shows cumulative HRV gains and attentional improvements lasting up to 3 weeks.
Upcoming VR‑VNS for Pain, cognition in psychosis, epilepsy, FND, extreme‑environment resilience
Multi‑centre grants in preparation
The Scientists Behind the Work
Our team brings together experts in neurostimulation, autonomic physiology, clinical research and data science.
We collaborate with leading academic and clinical partners to run controlled studies, analyse real-world data and refine stimulation protocols.
Together, we are advancing the science of closed-loop neuromodulation and turning it into practical, evidence-based solutions that improve lives.